- Blog
- Januvia free 30 day trial
- Disable mozilla keyboard shortcuts
- Endless space 2 tips vaulters
- How to update onenote 2016 drivers
- Os x yosemite vmware 3d acceleration
- Food network recipes pioneer woman mac and cheese
- Portal and portal 2 sales
- Html editor free download for windows 10 aptana or atom
- Hulu free 30 day trial
- Fatalities for mortal kombat for ps3
- Mac dvd player
- Smart card reader and writer software
- Cnet downloader free video converter download full version
- Where can i download books in pdf format
- Macphun luminar 2018 reviews
With the combination of Yanuvia with PPAR agonists, peripheral edema was often noted in patients. In the central nervous system, the most common adverse reactions were dizziness, cranialgia, and excessive drowsiness.įrom the gastrointestinal tract, when Yanuvia was admitted, there was dyspeptic syndrome, flatulence, stool disorders, discomfort in the upper abdominal cavity. Hypoglycemic conditions were most prevalent among patients who took Yanuvia with sulfonylureas - up to 14%, taking it against insulin therapy led to hypoglycemic conditions with a frequency of 9%. A combined treatment with Yanuvia with insulin was often accompanied by a flu.Īllergic reactions were noted with almost the same frequency in patients who took Yanugia, with monotherapy or as part of a two-three-component scheme. In the area of the respiratory tract in monotherapy Janow most common adverse reactions were infections of the upper parts of it, nasopharyngitis. Insulin-dependent type of diabetes and ketoacidosis are direct contraindications to the use of Yanuvia. The medicine is prohibited for use in children and pregnant women, during lactation. Yanuvia is not used in cases of excessive sensitivity to its constituents. The daily amount of Yanuvia - 100 mg is taken once per os, regardless of food, in combination with other hypoglycemic agents or under conditions of monotherapy. Januvia can be used against the background of insulin therapy with insufficient effectiveness of such therapy. In the triple scheme of therapy, Yanuvia is used in combination with sulfonylurea/metformin or metformin/agonist PPAR- γ, when the clinical effect of the double scheme is not enough.
#Januvia free 30 day trial Activator
Januvia is indicated as one of the components of combined therapy with metformin, sulfonylureas or PPAR- γ activator when there is no proper effect on glycemic control while dieting and moderate motor activity. The drug is prescribed to patients with ineffectiveness of diet therapy in combination with optimal physical activity.
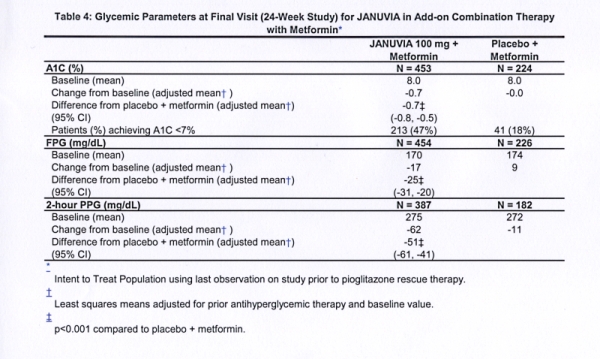
It is a highly selective inhibitor of DPP-4, the medicine has been widely used in the treatment of type 2 diabetes. With the combination of Yanuvia with PPAR agonists, peripheral edema was often noted in patients.Yanuvia belongs to the group of incretins.

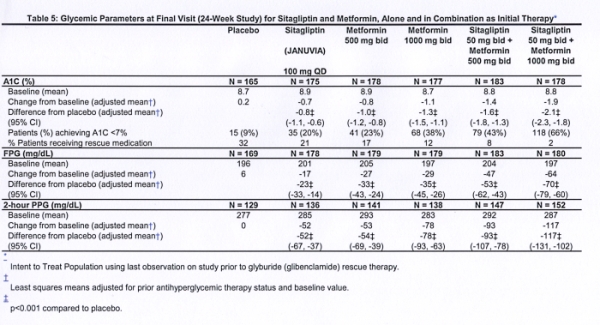
The coupon is valid for use once every 30 days with a qualifying prescription before the expiration date printed on the coupon. Patient is responsible for the first 5 of their out-of-pocket cost. Januvia is indicated as one of the components of combined therapy with metformin, sulfonylureas or PPAR- γ activator when there is no proper effect on glycemic control while dieting and moderate motor activity. The coupon is valid for up to 150 off your out-of-pocket cost on each qualifying prescription for JANUVIA, up to a 90-day supply per prescription fill. Yanuvia belongs to the group of incretins.
- Blog
- Januvia free 30 day trial
- Disable mozilla keyboard shortcuts
- Endless space 2 tips vaulters
- How to update onenote 2016 drivers
- Os x yosemite vmware 3d acceleration
- Food network recipes pioneer woman mac and cheese
- Portal and portal 2 sales
- Html editor free download for windows 10 aptana or atom
- Hulu free 30 day trial
- Fatalities for mortal kombat for ps3
- Mac dvd player
- Smart card reader and writer software
- Cnet downloader free video converter download full version
- Where can i download books in pdf format
- Macphun luminar 2018 reviews
